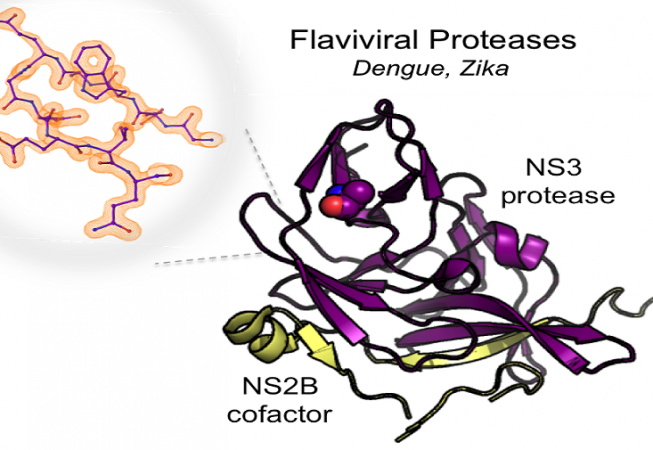
Flaviviruses, including dengue virus and Zika virus, contain a positive-sense RNA genome that is expressed as a single polyprotein precursor. Production of the active virus is dependent on the viral protease (NS2B-NS3pro) to cleave the polyprotein. As such, the protease is an excellent target to treat individuals who have been infected. Despite this knowledge, no protease-directed therapies have yet emerged, likely because most drug discovery efforts have focused on the active site. We are interested in studying allosteric sites on these viral proteases as an alternative way to inhibit their function. We have already identified a novel allosteric site centered at A125 that blocks the motion of a critical loop required for proteolytic activity. Our current research efforts are focused on discovering allosteric compounds that lock the enzyme into an inactive conformation and crystallizing such states to better understand the mechanism of inhibition.
Maureen E. Hill, Anil Kumar, James A. Wells, Tom C. Hobman, Olivier Julien, and Jeanne A. Hardy (2018). “The unique cofactor region of Zika virus NS2B-NS3 protease facilitates cleavage of key host proteins.” ACS Chemical Biology. pdf
Muslum Yildiz, Sumana Ghosh, Jeffrey A. Bell, Woody Sherman and Jeanne A. Hardy, (2013). “Allosteric inhibition of the NS2B-NS3 protease from dengue virus” ACS Chemical Biology. 8 (12): 2744–2752. pdf